11 December 2023
United States health officials last week approved two gene treatments for sickle cell disease. Doctors hope that the treatments will provide a cure for the painful genetic blood disorder.
The U.S. Food and Drug Administration (FDA) said the one-time treatments can be used for patients 12 and older with severe forms of the disease.
One treatment is made by Vertex Pharmaceuticals and CRISPR Therapeutics. The other is made by Bluebird Bio.
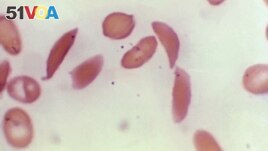
FILE - This photo provided on Oct. 25, 2023 by the Centers for Disease Control and Prevention shows crescent-shaped red blood cells from a sickle cell disease patient in 1972. (Dr. F. Gilbert/CDC via AP, File )
"Sickle cell disease is a... life-threatening blood disorder with significant unmet need," the FDA's Dr. Nicole Verdun said in a statement announcing the approvals. "We are excited to advance the field especially for individuals whose lives have been severely disrupted by the disease."
Sickle cell disease (or SCD) affects millions of people worldwide. The disease is common in places with high cases of malaria, like Africa and India. Scientists believe being a carrier of SCD helps protect against severe malaria.
It is more common among Black Americans, Africans, and Middle Easterners than other population groups. The World Health Organization said many children with the most severe form of the disease die before the age of five, usually from an infection or severe blood loss.
The new treatments
A genetic change, or mutation, causes blood cells to become defective in people with sickle cell disease. The unhealthy blood cells result in blockages, called blood clots. The clots cause pain and damage to bones and organs in the human body.
The Vertex treatment is called Casgevy. Its goal is to help the body go back to producing healthy blood cells that are present at birth. It uses CRISPR, the gene editing tool, to knock out a gene in stem cells collected from the patient.
Bluebird's treatment, called Lyfgenia, aims to add copies of a modified gene. The modified gene helps red blood cells to produce a protein that prevents or reverses unhealthy cells.
The two gene treatments are the first approved in the U.S. for sickle cell disease. Vertex is already approved in Britain and Bahrain.
The new treatments are costly. The cost for Bluebird Bio is $3.1 million and for Vertex, $2.2 million. To help cover the cost, the U.S. Centers for Medicare and Medicaid says it plans to create partnerships with state Medicaid agencies and drug companies.
Even with the high cost, many patients say they would consider the gene treatment because of the seriousness of the disease.
Jalen Matthews of Louisville, Kentucky, was diagnosed with sickle cell at birth. She had her first pain episode at age 9. Three years later, the disease led to a spinal cord stroke that left her with some paralysis in her left arm and leg.
"I had to learn how to walk again, feed myself, clothe myself, basically learn how to do everything all over again," said Matthews, who is now 26.
Today, she deals with the disease by having blood transfusions. About every two months, the treatment replaces five units of her blood with healthy cells. She said she plans to ask her doctor about the new treatments.
"This one-time kind of cure is very much needed," Matthews said.
Studies testing the treatments suggest they work well. Of 31 people treated in the Vertex study with enough follow-up, 29 were free of pain episodes for at least a year. In the Bluebird study, 28 of 32 patients had no severe pain or organ damage between six and 18 months after the treatment.
But doctors note there are possible side effects. They also say the long-term results are unknown. For both treatments, the required chemotherapy comes with risks such as infertility, hair loss and serious infections.
With the Bluebird treatment, blood cancer has happened. The FDA said it will include a "black box warning" about that risk.
With the Vertex therapy, some scientists worry that CRISPR brings the possibility of "off-target effects." These are unexpected changes to a person's genome.
I'm Anna Matteo.
Hai Do adapted this story for Learning English with reporting from The Associated Press and other sources.
_________________________________
Words in This Story
advance - v. to move forward
knock out - phrasal verb, to cause something to stop working
reverse - v. to return to an earlier state
episode - n. a short time of an illness
paralysis - n. a condition in which you are unable to move
blood transfusion - n. a medical treatment in which someone's blood is put into the body of another person